Burn Wound Microenvironment and Healing Prognosis
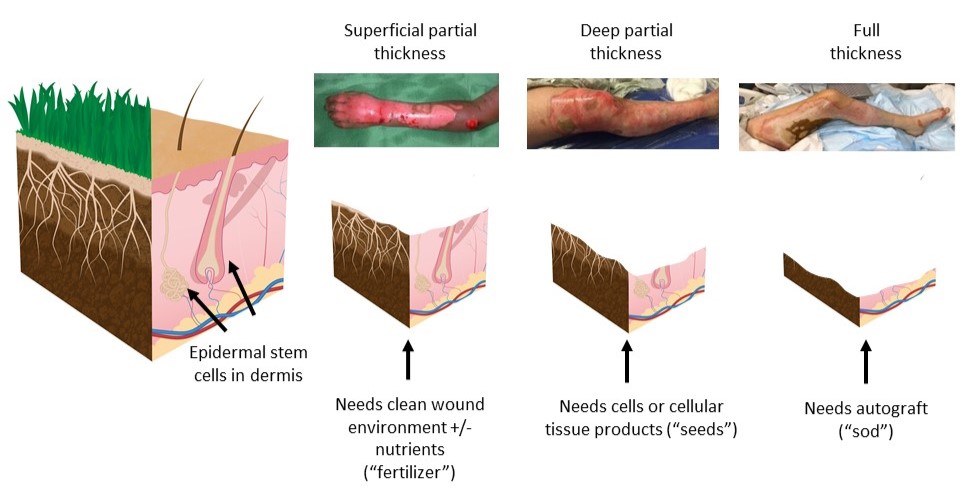
Burn injury that destroys tissue deep into the dermis often requires surgery to heal. However, the ability to determine the depth – and thus the regenerative capacity – of burn wounds is limited to visual assessment. Furthermore, the microenvironmental changes that occur over time have not been well studied in humans.
An analogy that is often helpful to understand the healing potential is if one considers a lawn as a model of skin. Each blade of grass starts deep in the soil in the root system of the lawn, similar to how the epidermal cells of the skin start in the hair follicles and sweat glands that originate deep in the dermis. When the top of the lawn is mowed, it grows back without any changes to the look of the lawn; this is similar to a superficial burn. When the lawn mower is set too low and some of the root system of the lawn is destroyed, the bare spots fill in with weeds instead of normal grass. In the skin, this happens if a deep partial thickness burn is allowed to heal over a long period of time without any surgical intervention; the bare spots are filled in with scar tissue, which can affect function. If the entire root system of the lawn is destroyed, it requires replacement with sod to replace the root system. This is analogous to a full thickness burn, which requires autologous skin grafting to replace the damaged skin.
Our lab is interested in understanding how to enhance healing in deep partial thickness wounds. There are gaps in knowledge around the exact amount of dermis and epithelial progenitor cells that are necessary to support autologous regeneration.
This is an accordion element with a series of buttons that open and close related content panels.
Relevant Publications
- Gibson ALF, Faucher LD. Is It the Holy Grail or Snake Oil? Ann Surg. 2025 Apr 1;281(4):559-560. doi: 10.1097/SLA.0000000000006556. Epub 2024 Oct 3. PMID: 39360408.
- Junak M, Gibson A. Burn Progression in Human Skin – A Review of Current Knowledge and Opportunities for Future Research. J Burn Care Res. 2025 Feb 17:iraf014. doi: 10.1093/jbcr/iraf014. Epub ahead of print. PMID: 39957288.
- Zajac J, Liu A, Hassan S, Gibson A. Mechanisms of delayed indocyanine green fluorescence and applications to clinical disease processes. Surgery. 2024 Aug;176(2):386-395. doi: 10.1016/j.surg.2024.03.053. Epub 2024 May 14. Review. PMID: 38749795; PMCID: PMC11246809.
- Zajac JC, Liu A, Uselmann AJ, Lin C, Hassan SE, Faucher LD, Gibson AL. Lighting the Way for Necrosis Excision Through Indocyanine Green Fluorescence-Guided Surgery. J Am Coll Surg. 2022 Nov 1;235(5):743-755. doi: 10.1097/XCS.0000000000000329. Epub 2022 Oct 17. PMID: 36102554; PMCID: PMC9753148.
- Schroeder AB, Karim A, Ocotl E, Dones JM, Chacko JV, Liu A, Raines RT, Gibson ALF, Eliceiri KW. Optical imaging of collagen fiber damage to assess thermally injured human skin. Wound Repair Regen. 2020 Nov;28(6):848-855. doi: 10.1111/wrr.12849. Epub 2020 Aug 21. PMID: 32715561; PMCID: PMC7755456.
- Barros-Becker, F., Squirrell, J.M., Burke, R., Chini, J., Rindy, J., Karim, A., Eliceiri, K.W., Gibson, A., Huttenlocher, A. Distinct tissue damage and microbial cues drive neutrophil and macrophage recruitment to thermal injury. iScience. 2020 Nov;23(11):101699. doi: 10.1016/j.isci.2020.101699. PMID: 33196024; PMCID: PMC7644964.
- Karim, A., Shaum, K., Gibson, A. Indeterminate depth burn injury — Exploring the uncertainty. J Surg Res. 2020 Jan;245:183-197. doi: 10.1016/j.jss.2019.07.063. Epub 2019 Aug 14. PMID: 31421361; PMCID: PMC8711117.
- Dones, J., Schroeder, A., Tanrikulu, I., Hoang, T., Gibson, A., Eliceiri, K., Raines, R. Optimization of interstrand interactions enables burn detection with a collagen-mimetic peptide. Org Biomol Chem. 2019 Nov 27;17(46):9906-9912. doi: 10.1039/c9ob01839e. PMID: 31720665; PMCID: PMC7018443.
- Miskolci V., Squirrell J., Rindy J., Vincent W., Sauer J.D., Gibson A, Eliceiri K.W., Huttenlocher A. Distinct inflammatory and wound healing responses to complex caudal fin injuries of larval zebrafish. Elife. 2019 Jul 1;8:e45976. doi: 10.7554/eLife.45976. PMID: 31259685; PMCID: PMC6602581.
- Karim AS, Yan A, Ocotl E, Bennett DD, Wang Z, Kendziorski C, Gibson ALF. Discordance between histologic and visual assessment of tissue viability in excised burn wound tissue. Wound Repair Regen. 2019 Mar;27(2):150-161. doi: 10.1111/wrr.12692. Epub 2018 Dec 26.|PMID: 30585657; PMCID: PMC6393178.
- LeBert, D., Squirrell, J., Freisinger, C., Rindy, J., Frecentese, G., Eliceiri, K.., Gibson, A., and Huttenlocher, A. Damage-induced reactive oxygen species regulates vimentin and dynamic collagen-based projections to mediate repair in larval zebrafish. Elife. 2018 Jan 16;7:e30703. doi: 10.7554/eLife.30703. PMID: 29336778; PMCID: PMC5790375.
- Gibson, A., Bennett, D., Taylor, L. Improving the histologic characterization of burn depth. J Cutan Pathol. 2017 Dec;44(12):998-1004. doi: 10.1111/cup.12991. Epub 2017 Jul 24. PMID: 28632906; PMCID: PMC9509661.
- Gibson ALF, Shatadal S. A simple and improved method to determine cell viability in burn-injured tissue. J Surg Res. 2017 Jul;215:83-87. doi: 10.1016/j.jss.2017.03.064. Epub 2017 Apr 7. PMID: 28688666.
- Israel, J., Greenhalgh, D., Gibson, A. Variations in burn excision and grafting: A survey of the American Burn Association. J Burn Care Res. 2017 Jan/Feb;38(1):e125-e132. doi: 10.1097/BCR.0000000000000475. PMID: 27893575.
Relevant Grants
Funding Agency & Program:
NIGMS R01GM145723
Dates:
07/01/2022 – 04/30/2027
Grant Title:
Fluorescence-based Detection of Inflammation and Necrosis to Inform Surgical Decision-Making and Enhance Outcomes
Funding Agency & Program:
NIH P30 – UW Skin Disease Research Center Pilot Grant
Dates:
12/01/2019 – 08/31/2021
Grant Title:
Novel Application of Indocyanine Green as a Biomarker to Identify Tissue Necrosis in Burn Patients
Funding Agency/Program:
Wisconsin Partnership Program – New Investigator Program
Dates:
07/01/2018 – 06/30/2020
Grant Title:
Autologous Regeneration in Burn Injured Patients
Funding Agency & Program:
NIH P30 – UW Skin Disease Research Center Pilot Grant
Dates:
09/01/2016 – 08/31/2017
Grant Title:
Molecular and Proteomic Signatures of Cellular Regeneration in Burn Tissue
Collaborators
Anna Huttenlocher, MD
Professor of Medical Microbiology & Immunology and Pediatrics
UW-Madison
Huttenlocher Lab
Di Yan, MD
Assistant Professor, Department of Dermatology
UW-Madison
Kevin Eliceiri, PhD
Professor of Medical Physics and Biomedical Engineering
UW-Madison
Eliceiri lab
Huy Dinh, PhD
Assistant Professor, Biostatistics and Medical Informatics, Oncology
UW-Madison
Dinh Laboratory
Commonly Utilized UW Core Facility Resources
Establishment of Human and Comparative Models for Wound Healing Research
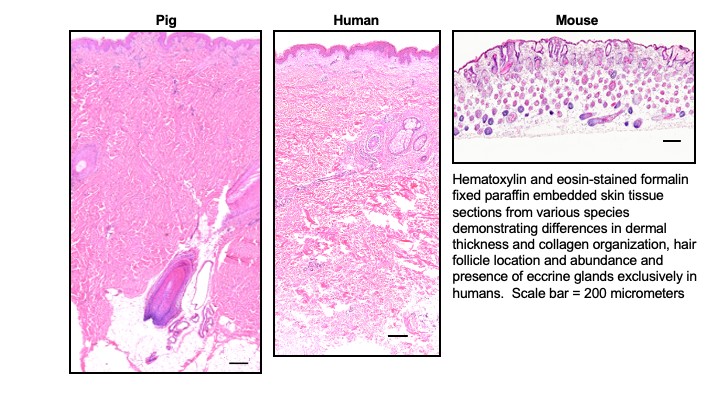
The challenges associated with translation of basic science discoveries in wound healing are partly rooted in the use of animal models, with wound healing and microanatomy that is vastly different than that of humans. To facilitate the translation of discoveries, our aim is to develop reproducible human burn models (ex vivo and in vivo) that simulate thermal injury sustained in patients to characterize human burn progression and healing. The models have provided an opportunity to enhance team science at our institution in the fields of engineering, microbiology, immunology, and advanced imaging.
This is an accordion element with a series of buttons that open and close related content panels.
Relevant Publications
- Liu A, Cheong JZA, Hassan S, Wielgat MB, Meudt JJ, Townsend EC, Shanmuganayagam D, Kalan LR, Gibson A. The effect of anatomic location on porcine models of burn injury and wound healing. Wound Repair Regen. 2024 May 22. doi: 10.1111/wrr.13190. Epub ahead of print. PMID: 38775411.
- Cheong JZA, Liu A, Rust CJ, Tran CL, Hassan SE, Kalan LR, Gibson ALF. Robbing Peter to Pay Paul: Chlorhexidine gluconate demonstrates short-term efficacy and long-term cytotoxicity. Wound Repair Regen. 2022 Sep;30(5):573-584. doi: 10.1111/wrr.13044. Epub 2022 Aug 11. PMID: 36638156; PMCID: PMC9542784.
- Gibson A, Liu A, Eliceiri K. Response to letter to the editor on “The use of human ex vivo models in burn research – Developments and perspectives”. Burns. 2021 Jun;47(4):968-969. doi: 10.1016/j.burns.2020.11.018. Epub 2020 Dec 11. PMID: 33934910.
- Liu A, Ocotl E, Karim A, Wolf JJ, Cox BL, Eliceiri KW, Gibson ALF. Modeling early thermal injury using an ex vivo human skin model of contact burns. Burns. 2021 May;47(3):611-620. doi: 10.1016/j.burns.2020.08.011. Epub 2020 Aug 29. PMID: 33279338.
- Gibson ALF, Carney BC, Cuttle L, Andrews CJ, Kowalczewski CJ, Liu A, Powell HM, Stone R, Supp DM, Singer AJ, Shupp JW, Stalter L, Moffatt LT. Coming to Consensus: What Defines Deep Partial Thickness Burn Injuries in Porcine Models? J Burn Care Res. 2021 Feb 3;42(1):98-109. doi: 10.1093/jbcr/iraa132. PMID: 32835360; PMCID: PMC7856457.
- Karim AS, Liu A, Lin C, Uselmann AJ, Eliceiri KW, Brown ME, Gibson ALF. Evolution of ischemia and neovascularization in a murine model of full thickness human wound healing. Wound Repair Regen. 2020 Nov;28(6):812-822. doi: 10.1111/wrr.12847. Epub 2020 Aug 8. PMID: 32686215; PMCID: PMC8592059.
- Moffatt LT, Madrzykowski D, Gibson ALF, Powell HM, Cancio LC, Wade CE, Choudhry MA, Kovacs EJ, Finnerty CC, Majetschak M, Shupp JW; Standards in Biologic Lesions Working Group. Standards in Biologic Lesions: Cutaneous Thermal Injury and Inhalation Injury Working Group 2018 Meeting Proceedings. J Burn Care Res. 2020 May 2;41(3):604-611. doi: 10.1093/jbcr/irz207. PMID: 32011688; PMCID: PMC7195554.
Relevant Grants
Funding Agency/Program:
Wisconsin Partnership Program New Investigator Program
Dates:
07/01/2018 – 06/30/2020
Grant Title:
Autologous Regeneration in Burn Injured Patients
Collaborators
Lindsay Kalan, PhD
Associate Professor, Biochemistry & Biomedical Sciences
McMaster University, Canada
Commonly Utilized UW Core Facility Resources
Novel Technology for Wound Healing
Translational research is necessary to ensure the transfer of basic scientific discoveries into meaningful patient benefit. Our work spans evaluation of human skin substitutes, advancing imaging techniques for clinical decision-making in burns, photodynamic therapy, and electrical stimulation bandage therapy for wound healing. Involvement with clinical research through investigator- and industry-sponsored research is necessary to make sure the right clinical needs and gaps in knowledge are being investigated.
This is an accordion element with a series of buttons that open and close related content panels.
Relevant Publications
- Liu, A., Ochoa, M., Reed, M., Junak, M., Pashaj, J., Pogue, B., and Gibson, ALF. “Indocyanine green and protoporphyrin IX fluorescence imaging of inflammation, hypoxia and necrosis of burns.” Burns & Trauma. Accepted Mar 2025.
- Zajac J, Liu A, Hassan S, Gibson A. Mechanisms of delayed indocyanine green fluorescence and applications to clinical disease processes. Surgery. 2024 Aug;176(2):386-395. doi: 10.1016/j.surg.2024.03.053. Epub 2024 May 14. PMID: 38749795; PMCID: PMC11246809.
- Holmes Iv JH, Gibson ALF, Short T, Joe VC, Litt J, Carson J, Carter JE, Wibbenmeyer L, Hahn H, Smiell JM, Rutan R, Wu R, Shupp JW. A phase 3b, open-label, single-arm, multicenter, expanded-access study of the safety and clinical outcomes of StrataGraft® treatment in adults with deep partial-thickness thermal burns. Burns. 2024 Jun 6:S0305-4179(24)00172-4. doi: 10.1016/j.burns.2024.05.023. Epub ahead of print. PMID: 39043513.
- Holmes Iv JH, Cancio LC, Carter JE, Faucher LD, Foster K, Hahn HD, King BT, Rutan R, Smiell JM, Wu R, Gibson ALF. Pooled safety analysis of STRATA2011 and STRATA2016 clinical trials evaluating the use of StrataGraft® in patients with deep partial-thickness thermal burns. Burns. 2022 Dec;48(8):1816-1824. doi: 10.1016/j.burns.2022.07.013. Epub 2022 Jul 28. PMID: 35941023.
- Zajac JC, Liu A, Uselmann AJ, Lin C, Hassan SE, Faucher LD, Gibson AL. Lighting the Way for Necrosis Excision Through Indocyanine Green Fluorescence-Guided Surgery. J Am Coll Surg. 2022 Nov 1;235(5):743-755. doi: 10.1097/XCS.0000000000000329. Epub 2022 Oct 17. PMID: 36102554; PMCID: PMC9753148. JACS “Talking Points” video.
- Liu A, Long Y, Li J, Gu L, Karim A, Wang X, Gibson ALF. Accelerated complete human skin architecture restoration after wounding by nanogenerator-driven electrostimulation. J Nanobiotechnology. 2021 Sep 20;19(1):280. doi: 10.1186/s12951-021-01036-7. PMID: 34544434; PMCID: PMC8454068.
- Gibson ALF, Smiell J, Yu TC, Böing EA, McClure EB, Merikle E, Holmes JH 4th. Determining clinically meaningful thresholds for innovative burn care products to reduce autograft: A US burn surgeon Delphi panel. Burns. 2021 Aug;47(5):1066-1073. doi: 10.1016/j.burns.2020.10.022. Epub 2020 Nov 9. PMID: 33303264.
- Gibson ALF, Holmes JH 4th, Shupp JW, Smith D, Joe V, Carson J, Litt J, Kahn S, Short T, Cancio L, Rizzo J, Carter JE, Foster K, Lokuta MA, Comer AR, Smiell JM, Allen-Hoffmann BL. A phase 3, open-label, controlled, randomized, multicenter trial evaluating the efficacy and safety of StrataGraft® construct in patients with deep partial-thickness thermal burns. Burns. 2021 Aug;47(5):1024-1037. doi: 10.1016/j.burns.2021.04.021. Epub 2021 Apr 23. PMID: 34099322.
- Gibson ALF, Smiell J, Yu TC, Böing EA, McClure EB, Merikle E, Holmes JH 4th. Response to Letter to the Editor “Defining a meaningful reduction of donor sites-Not as easy as it seems”. Burns. 2021 Jun;47(4):978. doi: 10.1016/j.burns.2021.01.020. Epub 2021 Mar 1. PMID: 33685811.
- Holmes JH 4th, Schurr MJ, King BT, Foster K, Faucher LD, Lokuta MA, Comer AR, Rooney PJ, Barbeau KF, Mohoney ST, Gibson ALF, Lynn Allen-Hoffmann B. An open-label, prospective, randomized, controlled, multicenter, phase 1b study of StrataGraft skin tissue versus autografting in patients with deep partial-thickness thermal burns. Burns. 2019 Dec;45(8):1749-1758. doi: 10.1016/j.burns.2019.07.021. Epub 2019 Aug 13. PMID: 31416637.
- Long Y, Wei H, Li J, Yao G, Yu B, Ni D, Gibson AL, Lan X, Jiang Y, Cai W, Wang X. Effective Wound Healing Enabled by Discrete Alternative Electric Fields from Wearable Nanogenerators. ACS Nano. 2018 Dec 26;12(12):12533-12540. doi: 10.1021/acsnano.8b07038. Epub 2018 Nov 29. PMID: 30488695; PMCID: PMC6307171.
Relevant Grants
Funding agency and program:
NIAMS 1R01AR084031
Dates:
2/27/2025 – 2/26/2029
Grant Title:
Enhanced Wound Healing Through Nanogenerator-Driven Self-Activated Electrical Stimulation
Funding Agency & Program:
Wisconsin Partnership Program – Collaborative Health Sciences Program
Dates:
09/01/2024 – 08/30/2027
Grant Title:
Investigating the Efficacy of Protoporphyrin-based Photodynamic Therapy in Burn Wound Healing in Porcine Models
Funding Agency & Program:
NIGMS R01GM145723
Dates:
07/01/2022 – 04/30/2027
Grant Title:
Fluorescence-based Detection of Inflammation and Necrosis to Inform Surgical Decision-Making and Enhance Outcomes
Funding Agency & Program:
UW2020
Dates:
05/01/2019 – 04/30/2022
Grant Title:
Nanogenerator-driven Self-activated Electrical Stimulation for Enhanced Wound Healing
Collaborators
Xudong Wang, PhD
Professor in Materials Science and Engineering
UW-Madison
Xudong Wang’s Nanoscience and Nanotechnology Group
Brian Pogue, PhD
Professor and Chair, Department of Medical Physics
UW-Madison
MOXI Lab
Kristyn Masters, PhD
Professor; Chair, Department of Bioengineering; Director, Center for Bioengineering
Bioengineering (CEAS)
University of Colorado
Masters Lab
Mark “Marty” Pagel, PhD
Professor
Department of Medical Physics
UW-Madison